Background: AlloHSCT induces long-term, cure in CAYA with malignant and non-malignant disorders, including those with primary immunodeficiencies (PID). However, post-transplant complications secondary to immunosuppression and slow or delayed immune reconstitution remains a cause of morbidity and mortality amongst AlloHSCT recipients (Satwani/Cairo, BBMT, 2009). Major complications include refractory viral infections, most importantly adenovirus (ADV), which is secondary to absent or decreased adenovirus specific T-cell immunity which occurs due to delayed immune reconstitution and/or continued immunosuppression (George/Cairo, BJH, 2012). The use of virus-specific (vs) CTLs in immunocompromised patients offer an opportunity to restore temporary virus specific immunity. From the Viral Cytotoxic T-Lymphocyte Consortium (VIRCTLC), we present safety and efficacy results on immune compromised, AlloHSCT, or SOT patients treated with familial ADV specific CTLs.
Objective: Determine the safety and efficacy of familial ADV specific CTLs for the treatment of refractory systemic ADV infections.
Design/Methods: Patient were deemed eligible if they had a history of AlloHSCT, SOT, or PID and had evidence of ADV infections refractory to anti-viral agents. Refractory ADV infection was defined by increasing serum RT-PCR DNA (by 1 log) after 7 days or persistent quantitative RT-PCR DNA copies after 14 days of appropriate anti-viral therapy, and/or known resistance/intolerance to anti-viral agents. Related donors were required to be matched ≥3 HLA (A, B, or DRB1) loci and have an adequate T-cell response to viral specific MACS ADV PepTivators®. We chose a direct selection method to facilitate more rapid generation of cellular products in a point of care setting (and with 14 hrs eligibility determination). Peripheral blood mononuclear cells were collected from eligible family donors using non-mobilized apheresis. vsCTLs were isolated using the
CliniMACS ® Prodigy following stimulation with viral specific MACS® GMP PepTivator® AdV5 Hexon, generously provided by Miltenyi Biotec®. ADV specific CTLs were enriched using a Cytokine Capture System (CCS). CD4+ and CD8+ vsCTLs were quantified by flow cytometry (Feuchtinger et al, Blood, 2010). The target cell dose was 0.5x10 4 CD3+ cells/kg (recipient weight) for haploidentical related donors and 2.5x10 4 CD3+ cells/kg for matched related donors. Repeated doses were permitted every 2 weeks to a maximum of doses in the absence of a complete response (CR) and adverse events. The following were used to define response: CR - undetected ADV PCR, partial response (PR) - at least one log decrease from baseline, progressive disease (PD) - at least one log increase from baseline, and patients with stable disease.
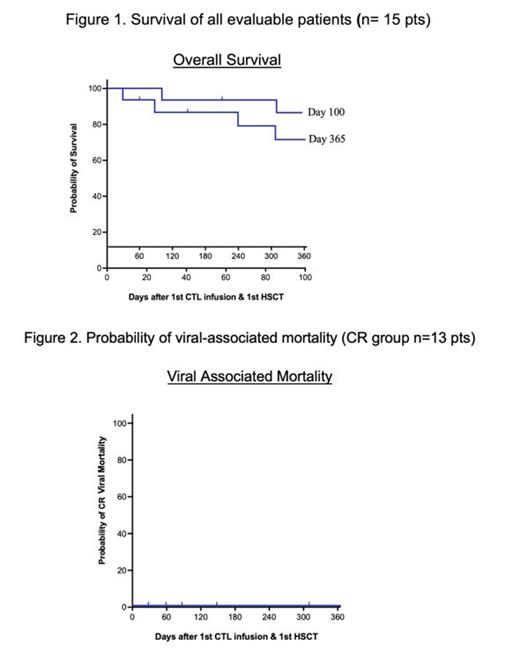
Results: Fifteen patients were enrolled: 10 females and 5 males; aged 1-19 years. All patients were post-AlloHSCT. All vsCTL donors were haploidentical: 8 were the original HSCT donors and 7 were third party donors (5 maternal, 2 paternal). The mean number of ADV CTL infusions was 3.7 (range: 1-16). One patient with underlying aplastic anemia had progressive disease complicated by increased adenoviral load despite 5 infusions of vsCTLs. Patient received FDA approval for additional CTLs, totaling 16 doses. Thirteen patients achieved CR as defined and 2 achieved PR. The overall response (OR) was 100% and the CR was 88%. The average time to OR was 33 days (range 6-112 days) and time to CR was 34 days (range 6-112 days). Day 100 and 365 overall survival (OS) post-HSCT was 86.1% (CI95: 49.4-95.7) and 70.5% (CI95: 38.9-87.8), respectively (Figure 1). For patients who achieved CR, the Day 100 OS post AlloHSCT was 83.9% (CI95: 55.0-94.3) and Day 365 post AlloHSCT was 74.6% (CI95: 39.8-91.1). Day 100 and 365 probability of ADV related mortality was 0% (Figure 2). One patient developed acute grade 2 skin GVHD possibly related to infusion, which resolved.
Conclusion: Our preliminary data demonstrate that haploidentical ADV specific CTLs manufactured by direct selections using the CCS with the CliniMACS® Prodigy and ADV specific peptivators are safe, well-tolerated, and efficacious in patients with refractory/persistent ADV infections after AlloHSCT. Manufacturing is rapid, reproducible, and effective. Accrual is ongoing. This research is supported by FDA RO1006301A1.
Disclosures
Lee:Avidicure B.V.: Consultancy, Current equity holder in private company, Research Funding; Kiadis Pharma, a Sanofi Corporation: Consultancy, Patents & Royalties: licensed through Nationwide Children's Hospital. Johnson:Miltenyi Biotech: Research Funding. Verneris:Allovir: Consultancy; Sanofi: Membership on an entity's Board of Directors or advisory committees; Qihan: Membership on an entity's Board of Directors or advisory committees; Jazz: Membership on an entity's Board of Directors or advisory committees; Medexus: Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Forge: Consultancy; Omeros: Consultancy. Dvorak:Alexion, AstraZeneca Rare Disease: Consultancy; Jazz Pharmaceuticals: Consultancy; Allovir: Consultancy. Cooke:Jazz Pharmaceuticals: Consultancy. Cairo:Jazz Pharmaceuticals: Consultancy, Honoraria, Research Funding, Speakers Bureau; Omeros Pharmaceuticals: Consultancy, Research Funding; Novartis: Consultancy; Servier Pharmaceuticals: Consultancy, Honoraria, Research Funding, Speakers Bureau; Amgen Inc.: Honoraria, Speakers Bureau; Sanofi: Honoraria, Speakers Bureau; Sobi: Honoraria, Speakers Bureau; Miltenyi Biotec: Research Funding; Merck: Research Funding; Astra Zeneca: Honoraria; Celularity: Research Funding; Abbvie: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal